Renal Replacement Therapy
Indications
Fluid removal
Acidosis
Uraemia (>30mmol/l)
Hyperkalaemia
OD of drug removed by RRT
Principals
Diffusion
Spontaneous migration of substances down concentration gradients due to random motion of molecules.
When liquid solvent passes through a membrane and the solute retained it is called osmosis.
Diffusion of solute (dissolved solid) across a membrane called dialysis.
In haemodialysis, diffusion across a semipermeable membrane is driven by solute concentration gradients between blood and dialysate fluid (crystalloid). Blood and dialysate fluid are pumped in opposite directions (counter current) in order to maximise the concentration gradient for diffusion.
Convection
Water is swept through a semi-permeable membrane down a pressure gradient (ultrafiltration). Any solutes in the water, that are small enough, also pass through (solvent drag or convection) much like in the glomerulus. Solute movement is driven by a pressure gradient rather than a concentration gradient. Filtration can be increased by increasing blood flow rate or increasing negative pressure in ultrafiltrate compartment (done by increasing the effluent flow pump rate).
Filtration pressure will be opposed by oncotic pressure and the relationship between the two will determine the filtration fraction.
In CVVH the positive pressure in the blood compartment is generated by a pump. The ultrafiltrate collects in bags and is discarded. Because so much water and electrolytes are lost for the necessary solute removal, these have to be replaced. The volume of replacement will depend on the fluid balance required for he patient.
Strictly speaking, the fluid removal that occurs (if desired) in IHD must be filtration but the mechanism of solute removal is dialysis.
Modes of RRT
Intermittent
Intermittent HaemoDialysis (IHD).
Continuous
Slow Continuous Ultrafiltration (SCUF). Used for slow fluid removal. There is no replacement fluid. Can be AV or VV.
Continuous Veno-Venous Haemofiltration (CVVH). Convective solute removal. Replacement fluid is infused at a rate depending on the desired overall fluid removal.
Continuous Veno-Venous Haemodialysis (CVVHD). Diffusive solute removal. Dialysate fluid is infused through the filter (along the outside of the fibres though which the blood flows) countercurrent to the direction of blood flow to maintain concentration gradients. Fluid can also be removed in this mode which is, of course, filtration.
Continuous Veno-Venous Haemodiafiltration (CVVHDF). Dialysis and convection are combined, again, with replacement fluid infused depending on desired fluid removal.
Hybrid techniques
Sustained Low Efficiency Dialysis (SLED)
Intermittent vs continuous
IHD
Typically performed for 3-5h x 3 a week. Used for outpatients with CRF but can also be used for ICU patients.
Rapid fluid and solute removal with significant fluid shifts.
Less popular than continuous techniques as perceived to be more CVS unstable (see evidence below).
CRRT
Continuous techniques are less efficient for solute removal but cause less haemodynamic changes. The membranes are more porous so are better for removing middle molecules such as cytokines and complement (inflammatory mediators) with potential theoretical benefits for sepsis (see evidence below). Cause cooling (but the circuit can be warmed). Avoids dysequilibrium syndrome (by slower solute removal and maintaining cerebral perfusion pressures).
Requires continuous anticoagulation, more work for nursing staff, more expensive.
Evidence consistently shows that the prescribed dose is rarely achieved due to interruptions such as filter clotting and trips to theatre and radiology.
Hybrid
Dose
Dose is strictly speaking solute clearance. This is difficult to measure so dose is usually described as the amount of effluent fluid in mls/kg/h:
CVVH - mls/kg/h of ultrafiltrate that is removed from the blood. On the machines I have used this is actually set by how much replacement fluid you dial in. This is assuming even balance. If aiming for negative balance the fluid loss has to be added to the replacement fluid rate to calculate dose/ultrafiltrate.
CVVHD - mls/kg/h of spent dialysis fluid.
CVVHDF - mls/kg/h of the combination of ultrafiltrate and spent dialysis fluid:
Effluent rate (ml/h) = patient fluid removal + any pre filter solution rate + post filter replacement solution rate + dialysate solution rate (all in mls/h).
Note that the replacement solution can be added pre or post filter or a combination of the 2. Either way, as in CVVH, the total replacement fluid is taken to be the same as the ultrafiltrate if even balance - you dial in ‘replacement’ rather than ‘ultrafiltrate’ for the ‘dose’ you want. Again you have to add in fluid removal to calculate ultrafiltrate if negative balance. Pre-dilution is counted in the effluent rate. As the concentration of unwanted solutes in the blood will be diluted, total solute clearance will be reduced thus lowering the ‘real’ dose and reducing the efficiency of the filter. Pre-dilution is unnecessary in CVVHDF as long as filtration fraction is kept below 30% with an adequate blood pump speed.
Solute removal in RRT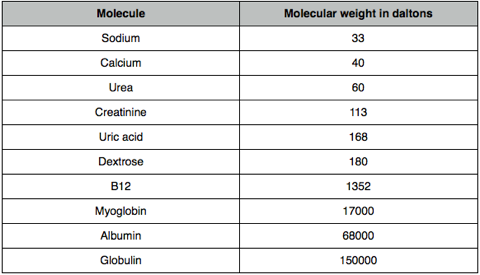
Filter pore sizes are around 30,000 daltons.
Molecules of low molecular weight (< 500 daltons) will pass freely through the filter; middle weight molecules (such as B12) will diffuse or convect but movement may be restricted; large molecules such as albumin will be retained.
You can work out from this what molecules do and don’t need to be replaced (in CVVH) or what molecules need to be in the dialysate fluid.
Drug removal in RRT
Un(protein)bound drug is removed and those with low MW (<500da)
Removed
Salicylates
Methanol
Ethylene glycol
Barbiturates
Lithium
Penicillins, Carbapenems, Aminoglycosides, Cephalosporins
Not removed
Digoxin
Tricyclic
Phenytoin
Benzodiazepines
B-blockers (atenolol is removed)
Metformin
Membranes
Cellulose
Used for dialysis
Poor permeability
Activate inflammatory cascades
Synthetic
High permeability (<20 kDa)
Less damage to PLT and WBC
CRRT membranes are all synthetic with high permeability.
Filters are made up of bundles of hollow fibres through which the blood flows. In dialysis these will be bathed by dialysate fluid.
This gives a huge surface area for diffusion or convection.
Coagulation
Filter clotting will cause failure of RRT.
All forms RRT activate clotting cascade.
Strategies to avoid filter clotting
Problems with filter clotting are nearly always a line problem rather than a circuit problem. The best site for a vascath is the right internal jugular vein as the line is straight rather than having to go round corners. Use the LIJ for normal central lines so the RIJ is left free for a vascath if needed.
Circuits are anti-coagulated with either heparin, citrate, prostacyclin (flolan) or lepiruden.
Anticoagulation is unnecessary if the patient’s INR or APPT ratio > 1.5 or PLT are < 50.
Heparin
The aim of heparin is to anticoagulate the circuit rather than the patient although some systemic anticoagulation will occur.
APPT is monitored to ensure that the patient is not over anticoagulated.
Can be monitored from samples both from the machine circuitry and from the patient.
Citrate
Gaining favour as no systemic anticoagulation.
Can cause hypocalcaemia.
Prostacyclin
Used to be used cases of suspected HIT which results in a prothrombotic state. It is a potent vasodilator reducing MAP around 15.
Lepiruden
A direct thrombin inhibitor now used for RRT in HIT. Monitoring is done by APTT.
Filtration fraction
A filtration fraction of <30% limits haemoconcentration. Increasing the blood pump speed will reduce the filtration fraction (aim for 20-30%) and make filter clotting less likely.
Predilution
Most CVVH or CVVHDF machines allow pre-dilution to reduce haemoconcentration. This reduces the efficiency of the filter and will lower the effective dose.
Pre-dilution is unnecessary in CVVHDF where the filtration fraction is already halved compared to CVVH (as half the dose comes from dialysis). An adequate blood pump speed will ensure a low enough filtration fraction - should be <30%.
How ultrafiltration dose (x-axis) and blood pump speed (y-axis) relate to filtration fraction. Blood pump speed should not be less than 150mls/min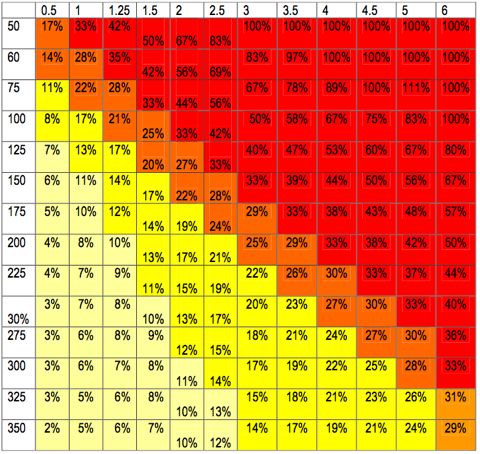
Site of line insertion
In order of preference:
RIJ, LIJ, subclavian, femoral
The RIJ will give you less filter problems as above.
The subclavian stenosis that results from vascath insertion is from long term lines. A few days of use on ICU should not cause any problems.
Femoral lines get infected most often and cause DVTs so should be a last resort.
Bicarb and lactate
HCO3 is freely filtered so needs replacing.
HCO3 containing mixtures have a short shelf life as they convert to carbonates which dissociate to CO2 which will diffuse out of the container wall.
Can also not be mixed with Ca or Mg as precipitate.
Lactate can be used as a substitute as it is metabolised to CO2 and H2O via the TCA cycle in the liver. A normal liver can metabolize 100 - 200 mmol/h of lactate. Caution in critical care due to either pre-existing high lactate from sepsis or poor metabolism from liver dysfunction.
HCO3 can either be infused separately or made up with replacement solution locally.
Sepsis
Most inflam mediators are middle sized (TNF, IL 1, 6, 8, PAF, complement) and removed by CVVH and CVVHDF.
Need ultrafiltration rate of 45ml/kg/h to significantly reduce plasma mediator concs because of their high generation rate.
Initial single centre evidence (Roncho) suggested high dose may improve mortality - see below.
Troubleshooting alarms - nomenclature depends on make/model
Negative access pressure
Patient moving or coughing
Access line against vessel wall - reposition line or swap lumens.
Disconnection.
Blood flow rate too high for the type of vascath.
Positive access pressure
Access line kinked or clotted
Blood flow rate too low for type of vascath.
Filter clotted
Clamped line
Replacement or pre-dilution fluid at too high a rate for the filter in use.
Clotted filter
Filter pressure positive / transmembrane pressure high
Clotting has begun to occur - pressure will rise gradually - check anticoagulation, line position, pre-dilution.
Kinked or clamped line - sudden pressure rise
Blood flow rate too high - high pressure from the start
Return pressure positive
Patient moving or coughing
Return line kinked or clamped
Vascath return lumen clotted or out of position.
Blood flow rate too high - high pressure from the start.
Return pressure low
Disconnection
Blood flow rate too low.
Timing, Dose and Mode - the evidence
RRT in patients with ARF. (systematic review)
JAMA 2008;299:793-805:
No differences in timing of starting it
No difference in intermittent or continuous
Cellulose membranes (rather than biocompatible membranes worse)
Benefit of high dose CRRT (35mls/kg/h) (did not include RENAL trial)
Intermittent vs continuous RRT
Cochrane Review 2007:
No difference in:
Mortality
Need for dialysis after discharge
LOS
Haemodynamic stability, occurrence of hypotension or escalation of vasopressors
CRRT had higher MAP
Conclusion
Choice of modality should be tailored to individual patient and unit
May be better to use CRRT in haemodynamically unstable but can still do intermittent safely
Intensity of RRT - RENAL investigators.
NEJM 2009;361:1627-1638:
1508 critically ill patients with AKI 40ml/kg/h vs 25
Actual doses 33 vs 22
Only difference was more hypophosphataemia in intensive group.
Delivered dose of RRT in critically ill patients with AKI
Vesconi et al. Crit Care 2009;13:R57:
553 patients. Observational. Looked at results and compared them with what dose RRT they had received. Intensive = >35mls/kg/h
No difference in mortality.
Shorter length of ventilation and stay in intensive group.
Patients in all trials get a significantly lower dose than that prescribed.
What should we use?
It doesn’t really matter whether you use intermittent or continuous or which continuous mode you choose.
No benefit of high dose (but it is not known whether a lower dose than used in the RENAL trial is worse) so aim for 25ml/kg/h (prescribe 30 as this will allow for interruptions).
Contrast induced nephropathy
Contrast causes renal ischaemia and direct tubular toxicity. Defined as increase in creat of 44 or >25% baseline within 72h of contrast.
Modern contrast solutions have a much lower osmolality than previously so less nephrotoxic.
Fluid loading, sodium bicarbonate, NAC and RRT have all been tried to prevent it.
RRT for prevention of contrast induced AKI: a meta-analysis.
Song et al Am J Nephrol 2010:
No benefit of prophylactic RRT to prevent CI-AKI.


